IVC Filter (inferior vena cava)Device used to catch blood clots
In 2010, the FDA issued a warning that IVC filters posed several significant risks including: filter fracture, device migration, and organ perforation. The FDA warning advised the filters should be removed immediately upon resolution of the underlying clot. This warning was updated in 2014 wherein the FDA advised the devices should be removed between the 29th and 54th day after implantation.
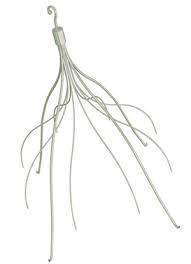
How do IVC filters work?
IVC (inferior vena cava) filters are small cone shaped medical devices that are implanted in the inferior vena cava. The device is usually placed just below the kidney to catch any blood clots that break loose from the deep veins in the lower legs. This device is routinely used as a treatment therapy for individuals suffering from DVT (deep vein thrombosis). The point is to catch the clot before it reaches the lungs (causing PE or pulmonary embolism), the heart (causing heart attack), or the brain (causing stroke). It is used as an alternative treatment therapy when more conservative treatments such as blood thinners fail to fully dissolve the clot. IVC filters are also used in individuals who are not candidates for blood thinning medications due to heredity or prior bleeding issues. Essentially, these filters are only intended to be used in high risk patients and as a treatment of last resort due to the many possible complications they can cause. It should be noted that long term use of these filters is not recommended and they should be removed when the clot and/or risk of clotting has resolved
Adverse Reactions from IVC Filters
The 2010 FDA warning was prompted after the filing of over 900 adverse event reports. When the legs of the filter fracture, the small metal pieces enter the blood stream and can cause significant damage to many major organ systems. Additionally, the complete device can break loose and migrate, in its entirety, throughout the circulatory system and cause even more severe complications including embolisms, organ perforation, heart attacks, strokes, internal bleeding, and oftentimes death.
People who currently have had and IVC placed or removed should be aware of the following potential side affects.
- Device migration
- filter fracture
- Embolization (movement of the entire filter or fracture fragments to the heart or lungs)
- Perforation of the IVC
- Difficulty removing the device
C.R Bard G2 and G2 Express Filters Sold thru 2010
Lawsuits against C.R. Bard IVC filters are being centralized in a Multi-District Litigation(No. 2641) in Arizona. Federal judges have 22 cases so far and is under U.S. District Judge David G. Campbell.
We want to hear from you
Speak with a representative now
Phone: (888) 430-5470
Email: [email protected]


